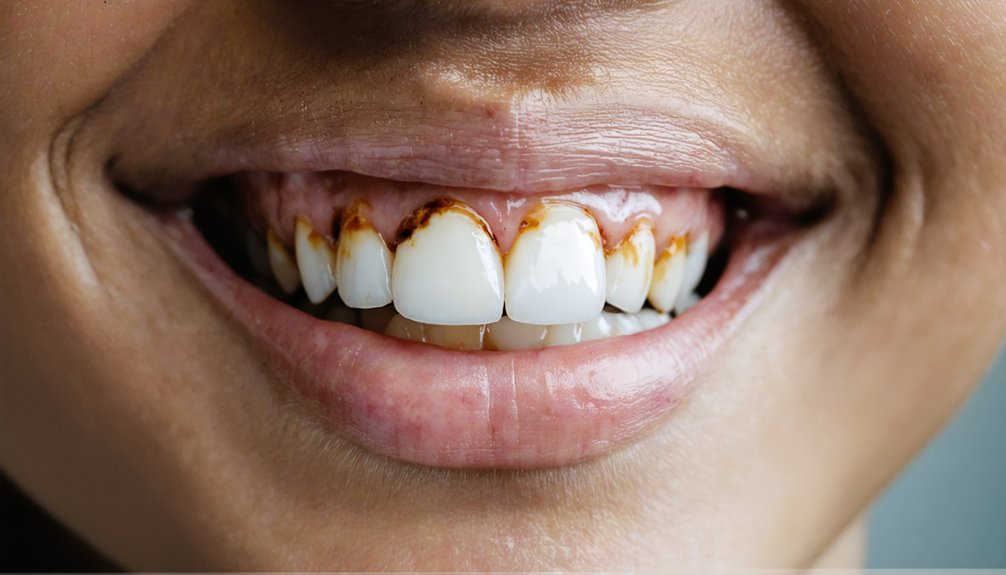
Fluoride-stained teeth resist whitening because excessive fluoride exposure during childhood permanently alters your enamel’s mineral structure at the crystalline level. This creates subsurface hypomineralized zones that trap discolored proteins deep within your enamel, where conventional peroxide-based bleaching agents can’t effectively penetrate. The fluoride forms irreversible fluorapatite crystals and causes intrinsic discoloration that’s chemically bonded to your tooth matrix rather than sitting on the surface. Understanding the specific mechanisms behind this resistance reveals why specialized treatments beyond standard whitening may be necessary.
Key Takeaways
- Fluoride permanently bonds with enamel to form fluorapatite crystals, creating irreversible structural changes that resist conventional whitening treatments.
- Excess fluoride creates hypomineralized zones within enamel that trap organic proteins and debris resistant to peroxide-based bleaching agents.
- Discoloration occurs deep within tooth structure rather than on surface, preventing standard whitening products from penetrating effectively.
- Chromophores forming white-to-brown stains are chemically bonded to the mineral matrix, making them difficult to oxidize or remove.
- Fluorapatite’s stronger crystal structure and trapped organic material require alternative treatments like microabrasion beyond traditional whitening methods.
What Causes Fluoride Stains During Tooth Development
Dental fluorosis occurs when developing teeth encounter excessive fluoride during a critical window from birth through age eight, permanently altering enamel structure before permanent teeth emerge. During tooth forming amelogenesis, your child’s ameloblast cells prove most vulnerable between ages 20-30 months.
Excessive fluoride intake—from swallowed toothpaste, water exceeding 1.5 ppm, or accumulated sources—triggers calcium signaling disruption within these enamel-producing cells. This interference causes mitochondrial dysfunction and endoplasmic reticulum stress, delaying amelogenin protein removal during the maturation phase. The result: hypomineralized, porous enamel that manifests as white opaque marks in mild cases or brown mottling in severe exposures.
Severity correlates directly with ingestion amount and duration. Once enamel development completes around age eight, fluoride exposure no longer causes fluorosis, but existing structural damage remains permanent.
How Fluoride Becomes Part of Your Tooth Structure
When fluoride ions enter your developing tooth structure, they chemically bond with hydroxyapatite crystals in the enamel’s mineral matrix, permanently replacing hydroxyl groups to form fluorapatite. This integration occurs at the crystal lattice level, creating a more acid-resistant mineral compound that becomes a fixed component of your tooth’s composition.
Once fluoride becomes incorporated into these crystalline structures during tooth formation, the resulting fluorapatite remains as a permanent part of your enamel throughout your lifetime.
Fluoride Binds to Hydroxyapatite
Understanding how fluoride integrates into your tooth structure requires examining the crystalline architecture of enamel. Your teeth contain calcium hydroxyapatite crystals with channels measuring 0.30–0.35 nm in diameter along their crystalline axis. The fluoride incorporation mechanism occurs when fluoride ions replace hydroxyl groups within these channels, fundamentally altering your enamel’s chemical composition.
This substitution transforms hydroxyapatite into fluorapatite through cycles of demineralization and remineralization. The fluoride binding affinity proves stronger than hydroxyapatite’s original structure because fluoride ions create more stable chemical bonds with calcium ions. Unlike topical hydroxyapatite particles that attach to your tooth surface, fluoride chemically modifies existing crystals from within. This integration produces fluorapatite, which demonstrates superior acid resistance and increased hardness compared to your natural hydroxyapatite—making fluoride-altered enamel structurally different from your original tooth composition.
Forming Permanent Fluorapatite Crystals
During enamel development, fluoride ions chemically replace hydroxyl groups (OH-) within your tooth’s hydroxyapatite crystals, creating fluorapatite through a permanent substitution reaction. This exchange produces crystal structure modifications that fundamentally alter your enamel’s physical properties.
The transformation occurs through three mechanisms:
- Ion substitution – Fluoride ions permanently occupy channels within the hexagonal crystal lattice, displacing hydroxyl groups along the c-axis
- Volume reduction – The substitution decreases crystal volume while simultaneously increasing structural stability and acid resistance
- Enhanced remineralization kinetics – Free fluoride ions accelerate mineral deposition, strengthening the crystalline matrix
Once incorporated, fluorapatite becomes an irreversible component of your tooth structure. This stability explains why fluoride-induced discoloration resists conventional whitening treatments—you’re attempting to alter permanently modified crystals rather than surface stains.
Integration Into Mineral Matrix
The mineral matrix of your developing tooth enamel contains hexagonal hydroxyapatite crystals with structural channels that accommodate specific ions, including calcium (Ca²⁺) and hydroxyl (OH⁻) groups. When you’re exposed to fluoride during tooth development, these ions undergo substitution within the crystal lattice—fluoride replaces hydroxyl groups to form fluorapatite (Ca₁₀(PO₄)₆F₂). This exchange permanently alters your developing enamel structure at the molecular level.
Post-eruption, the remineralization process allows topical fluoride to integrate into surface crystals, creating an acid-resistant layer. However, excessive fluoride during enamel maturation disrupts protein removal and mineral deposition, leaving wider crystalline gaps. These structural spaces retain organic material and increase porosity, creating permanent pathways for pigment penetration. You can’t reverse this integration—fluoride becomes a fixed component of your enamel’s mineral matrix.
Why Fluorosis Creates Intrinsic Discoloration
When excess fluoride disrupts ameloblast function during tooth development, it creates subsurface hypomineralized zones within your enamel matrix. These porous areas permanently trap organic proteins and cellular debris that form chromophores, resulting in the characteristic white-to-brown discoloration you observe.
Unlike extrinsic surface stains, this structural incorporation of pigmented molecules occurs deep within your enamel and dentin, making the discoloration intrinsic and resistant to conventional whitening methods.
Fluoride Binds Enamel Structure
As fluoride ions integrate into your developing tooth enamel, they permanently replace hydroxyl groups in the hydroxyapatite crystal lattice to form fluorapatite. While fluorapatite strengthens structural integrity against acid dissolution, excessive fluoride during maturation creates crystal flaws that compromise your enamel’s appearance.
This disruption manifests through three key mechanisms:
- Protein retention – Fluoride inhibits matrix proteinases, preventing proper removal of amelogenin fragments and restricting crystal growth
- Increased porosity – Defective crystallization creates microscopic gaps that trap discoloration intrinsically within your enamel structure
- Permanent integration – Fluoride bonds electrostatically within the crystal lattice, making structural defects irreversible through conventional surface treatments
These dose-dependent effects occur when plasma fluoride levels reach 2-12 µmol/L during tooth development, permanently altering your enamel’s mineral composition.
Organic Debris Traps Permanently
During enamel maturation, fluoride-disrupted ameloblasts trap amelogenin fragments and non-degraded matrix proteins directly within your developing tooth structure. This organic material undergoes permanent encapsulation as hypomineralized enamel solidifies around it, creating intrinsic stains that resist standard whitening approaches.
You’re facing difficult extraction challenges because peroxide-based bleaching agents can’t penetrate deeply enough to oxidize debris locked within enamel layers. The organic compounds remain chemically bonded to your tooth’s mineral matrix throughout its lifetime. As years pass, this trapped material gradually darkens from white opacity to brown discoloration through oxidation.
Your whitening options require alternative strategies: sodium hypochlorite applications can oxidize chromogenic compounds within accessible enamel, while microabrasion mechanically removes affected superficial layers. Severe cases necessitate restorative coverage through bonding or veneers.
The Science Behind Whitening Resistance in Fluoride-Stained Teeth
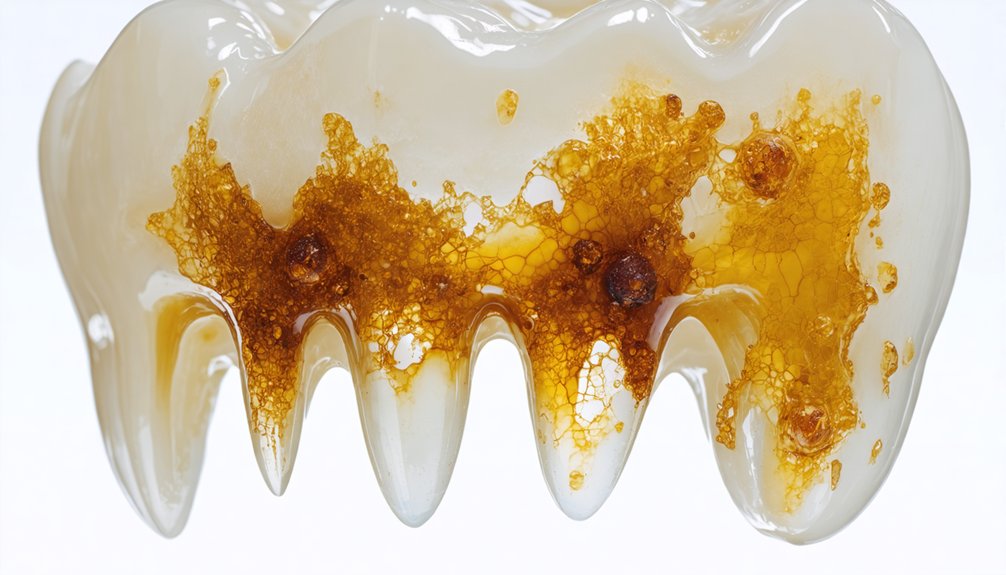
Fluoride-stained teeth resist conventional whitening treatments due to fundamental structural alterations within the enamel itself. When you’ve experienced excessive fluoride exposure during tooth development, the resulting hypocalcification creates permanent discoloration that bleaching agents can’t effectively address.
Three key mechanisms explain this whitening resistance:
- Crystal matrix disruption embeds stains deep within your enamel’s subsurface layers, placing them beyond the reach of peroxide-based treatments
- Remineralization limitations prevent fluorotic zones from responding to strengthening protocols that benefit healthy enamel
- Dehydration effects from bleaching temporarily accentuate the contrast between hypomineralized and normal areas, making stains more visible post-treatment
Your fluoride-stained enamel exhibits increased porosity and altered prism structure that hydrogen peroxide penetrates but can’t fundamentally change, leaving intrinsic discoloration intact.
Why Traditional Bleaching Agents Struggle With Fluoride Stains
Traditional bleaching agents fail against fluoride stains because the discoloration exists in chemically altered enamel that resists oxidation. Hydrogen peroxide concentrations (5-35%) show insufficient chromogen breakdown in fluorosis lesions, as hypermineralized structures trap organic material within porosities that evade peroxide penetration.
Your enamel’s protein-coated crystallite surfaces block oxidant diffusion, preventing staining chromogen breakdown even with extended application times. Carbamide peroxide demonstrates highest efficiency among conventional agents, yet ΔE differences remain non-significant compared to untreated fluorotic enamel. Sodium hypochlorite requires 25-30 minutes continuous contact but leaves persistent yellow discoloration.
Without enamel porosity mitigation through acid etching and resin sealing, degraded chromogens re-enter porous structures. McInnes solution (36% HCl, 30% H₂O₂) outperforms peroxide-alone protocols by addressing both structural obstruction and chromogen oxidation simultaneously.
When Professional Whitening Works for Fluorosis
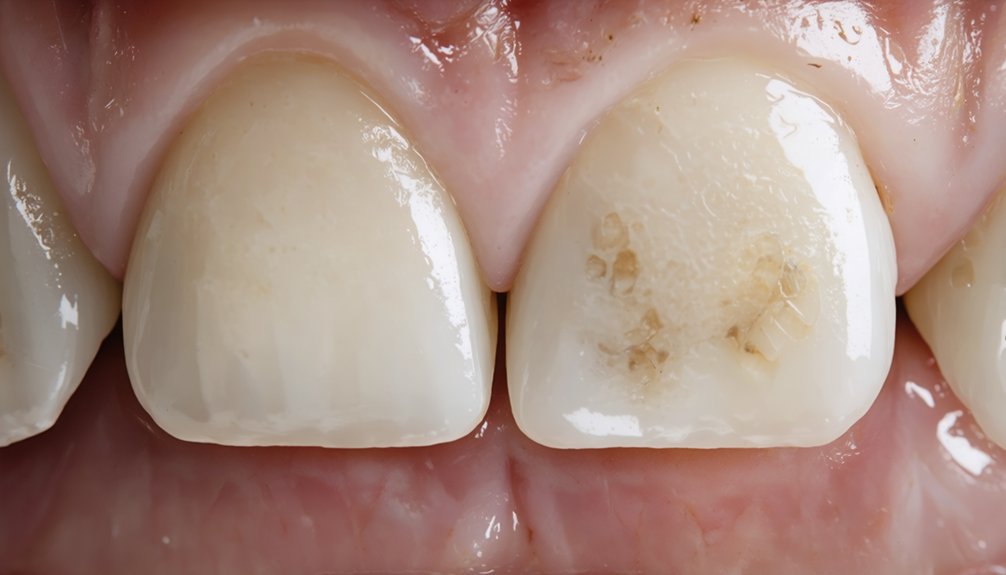
When fluorosis presents as mild white spots or faint streaking, professional peroxide-based whitening can effectively reduce the contrast between stained and unaffected enamel. Your dentist administers higher peroxide concentrations that penetrate deeper than over-the-counter products, with effective gum protection and clinical monitoring throughout treatment.
Professional whitening treatments use concentrated peroxide formulas to minimize the appearance of mild fluorosis under expert dental supervision.
Specialized protocols optimize outcomes for fluorosis patients:
- Customized whitening trays guarantee even peroxide distribution across affected areas, preventing patchy results
- Refrigerated gel formulations like KӧR maintain hydrogen peroxide potency while minimizing enamel dehydration that makes fluorosis more visible
- Post-treatment fluoride applications solidify remineralized enamel and extend brightness duration
Professional supervision allows your dentist to assess stain depth, select appropriate peroxide concentrations, and modify protocols based on your response. Single in-office sessions deliver dramatic improvements for eligible mild cases, though intrinsic hypomineralization beneath enamel may resist complete removal.
Advanced Bleaching Techniques That Penetrate Deeper
For moderate-to-severe fluorosis cases where standard whitening falls short, your dentist can employ specialized bleaching protocols that penetrate hypomineralized enamel layers. In office bleaching advancements include sodium hypochlorite application after phosphoric acid etching, allowing oxidizing agents to reach chromogenic material trapped within porous enamel.
High-concentration hydrogen peroxide gels—activated by LED or laser light—release oxygen molecules that break down intrinsic dentin stains in 30-60 minute sessions, achieving 2-8 shade improvements. Refrigerated carbamide peroxide formulations maintain stability for prolonged penetration during custom tray treatments.
Fluorosis whitening synergies emerge when combining microabrasion with professional bleaching, addressing both surface and deeper discoloration simultaneously. These targeted approaches require dentist supervision to guarantee safe application on matured enamel while maximizing stain degradation in hypomineralized zones resistant to conventional methods.
Alternative Treatment Options Beyond Whitening

While professional bleaching addresses many fluorosis concerns, persistent or severe discoloration often requires cosmetic restorations that physically mask affected enamel rather than chemically alter it.
Cost effective options include:
- Dental bonding – Tooth-colored resin covers discolored areas, shapes naturally, and lasts four to eight years. It serves as an economical substitute for more expensive restorations.
- Resin infiltration – This non-invasive method targets early-stage white spots through superficial enamel porosification and resin injection, requiring no drilling while providing immediate improvement.
- Remineralization therapy – Professional-grade calcium phosphate gels reduce white stains by promoting natural enamel repair without invasive procedures.
For comprehensive aesthetic enhancements, porcelain veneers deliver superior stain resistance and ten-to-fifteen-year longevity. Enamel microabrasion removes superficial stains while preserving tooth structure, often combined with whitening for ideal results.
Frequently Asked Questions
Can Fluoride Staining Be Prevented Once Permanent Teeth Have Already Erupted?
No, you can’t prevent fluoride staining once permanent teeth have erupted. However, you can maintain ideal fluoride intake levels through professional fluoride application methods to strengthen enamel without risking additional fluorosis on already-emerged teeth.
Do Electric Toothbrushes Remove Fluoride Stains Better Than Manual Brushing?
Electric toothbrushes remove 21% more plaque but won’t eliminate fluoride stains—they’re intrinsic, not surface-level. While you’ll gain improved plaque removal and better reach between teeth, professional whitening treatments remain your only effective option for fluoride-stained enamel.
Will My Children Develop Fluorosis if They Use Fluoride Toothpaste?
Your children won’t develop fluorosis if you follow proper fluoride dosage guidelines: rice-grain amounts under age three, pea-sized portions until six, supervised brushing, and regular dental checkup frequency. You control their fluoride exposure through appropriate supervision.
How Long Does Kör Deep Bleaching Treatment Typically Take to Complete?
You’ll complete KöR deep bleaching in 2-6 weeks: two weeks of nightly at-home treatment, followed by one in-office session. This typical treatment duration delivers expected results of up to 16 shades lighter, even for fluorosis-stained teeth.
Are There Specific Foods That Worsen the Appearance of Fluoride Stains?
Yes, acidic foods and dark-colored beverages can intensify fluoride stains’ visibility. You’ll notice increased discoloration when consuming coffee, red wine, berries, and tomato-based products, as they deposit additional pigments onto already-compromised enamel affected by fluorosis.