When you apply peroxide-based whitening agents to your teeth, they decompose and release free radicals—unstable molecules with unpaired electrons that penetrate your enamel through interprismatic spaces. These radicals target chromophores, the complex organic molecules causing discoloration in your dentin and enamel, breaking their conjugated double bonds through oxidation. This chemical reaction fragments large pigmented molecules into smaller, colorless compounds that no longer absorb light, effectively whitening your teeth. The following sections explain the complete oxidative process, ideal treatment conditions, and safety considerations you’ll need to understand.
Key Takeaways
- Peroxide bleaching agents decompose into free radicals that penetrate enamel, reaching discolored dentin within 15 minutes of application.
- Free radicals attack chromophore molecules by breaking conjugated double bonds, fragmenting large pigmented compounds into smaller colorless particles.
- Perhydroxyl radicals form in alkaline conditions (pH 9.5-10), providing optimal reactivity to oxidize stain molecules effectively.
- The oxidation process converts light-absorbing pigments into simpler molecular structures that no longer cause visible tooth discoloration.
- Professional formulations maintain pH between 4.5-5.5 to maximize whitening efficacy while protecting enamel from demineralization damage.
The Science Behind Peroxide-Based Bleaching
When hydrogen peroxide contacts tooth enamel, it initiates a chemical oxidation process that fundamentally alters chromophore molecules responsible for discoloration. The peroxide penetrates your enamel through interprismatic spaces, reaching the underlying dentin within 15 minutes.
There, free radicals—particularly perhydroxyl (HO₂) at pH 9.5-10—target double bonds in stain molecules, breaking them into smaller, colorless particles through electron transfer.
This oxidation addresses dentin discoloration effects specifically, as peroxides diffuse through enamel to interact with organic chromophores in deeper tissue layers. The process increases lightness by 19.9 ± 6.5° compared to alternative treatments.
You’ll notice surface texture changes minimally affect the enamel’s inorganic structure, while reactive oxygen molecules systematically dismantle stain chains. Professional concentrations (35-40% hydrogen peroxide) optimize this controlled oxidative reaction.
Breaking Down Hydrogen Peroxide and Carbamide Peroxide
When you apply peroxide-based whitening agents to your teeth, hydrogen peroxide undergoes immediate decomposition through exposure to light, heat, or salivary enzymes, releasing highly reactive oxygen radicals.
Carbamide peroxide breaks down into approximately one-third hydrogen peroxide and two-thirds urea, with the urea component elevating pH to 9.5-10 and promoting formation of perhydroxyl ions (HO₂⁻)—the most potent free radicals for bleaching. These free radicals penetrate enamel interprismatic spaces within 15 minutes, initiating electron transfer from chromophore molecules and disrupting the chemical bonds responsible for tooth discoloration.
Chemical Decomposition Process
Several distinct chemical reactions govern how peroxide-based whitening agents break down to produce their bleaching effects. When you apply carbamide peroxide, it decomposes into hydrogen peroxide and urea, releasing approximately 33% hydrogen peroxide by weight. The hydrogen peroxide then undergoes radical generation, forming hydroxyl (HO•) and perhydroxyl radicals as intermediary products before breaking down into water and nascent oxygen.
These radicals represent the rate-limiting step in decomposition. Temperature elevation of 10°C increases decomposition rates by a factor of 2.2, while metal ions and blue light irradiation accelerate this process. The byproduct effectiveness stems from oxygen molecules reacting directly with discoloration pigments. Research confirms this oxidative mechanism preserves enamel integrity—treating teeth for four days produced significant whitening (p<0.05) without demineralization or altered organic-inorganic ratios.
Free Radical Formation
The molecular instability of peroxide compounds drives their effectiveness as bleaching agents through a cascade of free radical generation. When you apply hydrogen peroxide at concentrations of 35-40%, it decomposes into water and oxygen while producing highly reactive species.
In alkaline conditions at pH 9.5-10, perhydroxyl (HO2) radicals form with ideal radical stability for enhanced bleaching outcomes.
Carbamide peroxide offers you controlled decomposition, releasing hydrogen peroxide gradually over eight hours with peak activity in the first two hours. These free radicals possess unpaired electrons, creating inherent instability that translates to exceptional chromogen reactivity. They oxidize pigmented molecules by breaking conjugated double bonds and stealing electrons, fragmenting complex stain structures into simpler, colorless compounds throughout your enamel and dentin layers.
Optimal Ph Conditions
Maintaining pH between 4.5 and 5.5 maximizes your teeth whitening efficacy while protecting enamel integrity from acid-induced demineralization. This slightly acidic environment activates hydrogen peroxide’s bleaching mechanism without excessive enamel erosion.
Professional formulations employ buffering system design to stabilize peroxide while preventing pH drops below 4.5, which would liberate calcium and phosphate ions from your enamel surface.
Carbamide peroxide products utilize urea as a stabilizer, controlling hydrogen peroxide release over extended periods. While 10% carbamide peroxide yields 3.6% hydrogen peroxide gradually, this slower breakdown necessitates longer exposure times.
You’ll achieve preferred results through pH control measures that maintain acidic conditions sufficient for peroxide stability (below 7.0) yet prevent aggressive demineralization. FDA-approved concentrations combined with neutral-pH formulations reduce sensitivity while delivering effective whitening outcomes.
How Free Radicals Transform Stain Molecules
When hydrogen peroxide breaks down within tooth structures, it sets off a cascade of oxidative reactions that fundamentally alter stain molecules at the molecular level. Free radicals target chromophores—pigmented compounds responsible for tooth discoloration—initiating chain cleavage reactions that fragment large organic molecules into smaller, colorless components.
These radicals attack conjugated double bonds within chromogens, converting them to single bonds through oxidation. This transformation reduces light absorption capacity, directly diminishing visible staining. Chromogen rearrangements occur as structural modifications simplify complex pigment molecules. Epoxidation affects conjugated systems, while ketone-containing stains undergo Baeyer-Villiger oxidation.
You’ll observe that peroxide itself remains inactive; the highly reactive radicals perform the actual oxidation work. This chemical transformation happens in situ—stains don’t physically detach but undergo irreversible molecular changes that produce your whitening effect.
Types of Tooth Discoloration That Respond to Oxidation
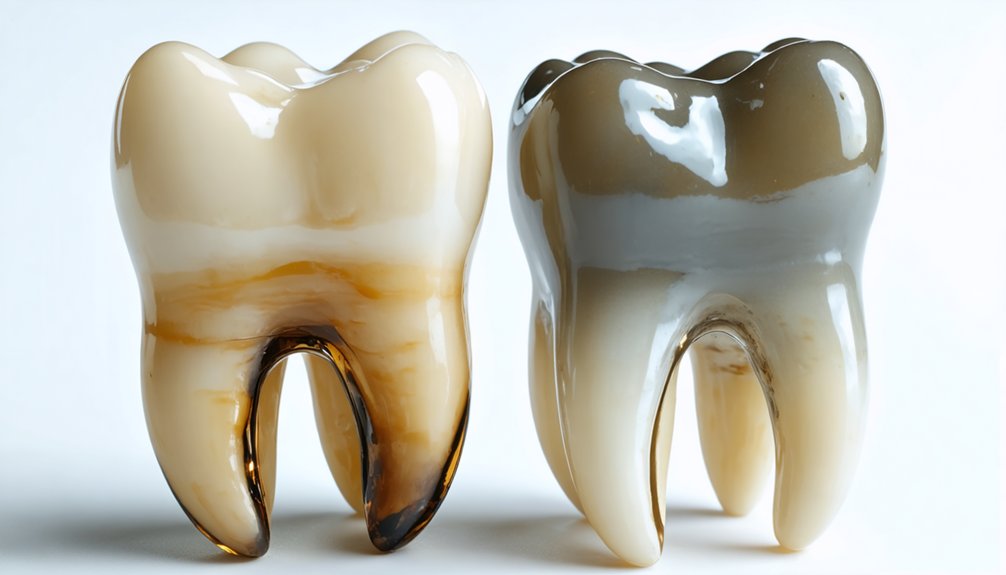
Understanding these oxidative mechanisms reveals their clinical application across distinct categories of tooth discoloration. Understanding intrinsic discoloration from tetracycline antibiotics requires extended peroxide exposure to oxidize chromophores embedded within your enamel and dentin structures.
Fluorosis-related staining responds as peroxide oxidizes altered enamel matrices, converting brown chromogenic deposits to colorless compounds. Addressing age-related yellowing involves penetrating the enamel-dentin interface where secondary dentin deposits and accumulated staining agents reside.
Extrinsic stains from coffee, tobacco, and dietary chromogens respond directly when peroxide contacts surface-level discoloration. Porphyrin-based stains particularly benefit from oxidative treatment, as iron within these compounds catalyzes peroxide reactions. Medication-induced discoloration, including eugenol-zinc oxide complexes, undergoes degradation through hydrogen peroxide’s chromophore-targeting oxidation, enabling you to manage diverse discoloration etiologies systematically.
The Journey of Peroxide Through Enamel and Dentin
Upon application to tooth surfaces, hydrogen peroxide molecules initiate penetration through enamel interprismatic spaces, creating pathways toward the underlying dentin. Your chosen concentration—ranging up to 40% in professional treatments—directly determines permeation speed, with complete nerve reach occurring within 5-15 minutes.
Once peroxide reaches the enamel-dentin junction, it diffuses into dentin regions where decomposition generates perhydroxyl free radicals at pH 9.5-10. These oxygen radicals target the dentin organic matrix, establishing distinct enamel oxidation patterns while addressing dentin matrix discoloration through chromogen breakdown.
The oxidation process converts high molecular weight colored compounds into smaller, lighter molecules, achieving lightening of 19.9 ± 6.5° in sound teeth. Oxygen persists within tooth structures for up to two weeks post-application, ensuring sustained whitening effects without altering enamel’s mineral-to-organic content ratios.
Professional vs. At-Home Whitening Methods
Choosing between professional and at-home whitening methods hinges on your desired outcome timeline, budget constraints, and tolerance for concentration-dependent peroxide exposure. In-office treatments deliver 8-10 shade improvements in 60-90 minutes using up to 35% hydrogen peroxide, maintaining results for 1-3 years.
Cost accessibility trade offs become evident when comparing $500-1,000 professional sessions against $20-50 over-the-counter strips that lighten teeth 1-2 shades over 14 days. Professional take-home kits with custom trays bridge effectiveness differences, providing superior gel concentrations and even coverage compared to generic two-dimensional strips.
Your selection should account for peroxide concentration’s direct correlation with whitening speed and durability. Maintenance products containing abrasive rather than oxidative components offer minimal bleaching but preserve existing brightness between professional interventions.
Changes in Tooth Appearance After Oxidative Treatment
When oxidative whitening agents penetrate your enamel, they fundamentally alter how your tooth structure interacts with visible light through changes in absorption, reflection, and scattering patterns.
Your teeth display enhanced fluorescence as oxidized chromophores reduce light absorption in the yellow spectrum while increasing blue-light reflection. These optical changes occur alongside temporary demineralization effects that modify enamel’s crystalline structure, contributing to the immediate post-treatment brightness you’ll observe.
Altered Light Absorption Properties
As hydrogen peroxide penetrates the enamel matrix, free radicals systematically disrupt the conjugated electron systems within chromogenic molecules, fundamentally altering their light absorption characteristics. You’ll observe measurable reductions in wavelength absorption as oxidation converts complex chromophores into simpler, stable compounds. This molecular transformation directly correlates with reduced chromogen adsorption within your enamel structure.
The oxidative breakdown you’ve initiated changes how light interacts with your tooth surface. Modified electron configurations diminish the molecules’ capacity to absorb incident light, while light refraction changes occur as simplified structures scatter photons differently than original stain compounds. Your teeth appear brighter because treated enamel reflects more light rather than absorbing it. Spectrophotometric assessment quantifies these changes, demonstrating proportional increases in reflectance corresponding to chromogen elimination degree. You’re controlling tooth brightness through calculated molecular simplification.
Enhanced Fluorescence and Scattering
Beyond absorptive modifications, oxidative whitening fundamentally restructures your tooth’s optical behavior through enhanced fluorescence and light scattering mechanisms. Post-treatment fluorescence intensity increases at the 440 nm emission peak as oxidation alters endogenous fluorophores within your enamel’s organic matrix. Quantitative light induced fluorescence devices detect these structural modifications, confirming treatment efficacy through measurable spectroscopic changes.
Simultaneously, oxidative processes reduce dentine opacity and enhance peritubular dentine deposition, intensifying light scatter throughout tooth structures. This scattering amplification produces chromatic saturation changes that contribute considerably to perceived whiteness under clinical lighting conditions. Your teeth demonstrate improved translucency as scattered light interacts differently with modified calcium apatite matrices.
These combined fluorescence and scattering enhancements enable superior esthetic masking post-whitening, ensuring restored teeth integrate seamlessly with your natural dentition under both UV and ambient illumination.
Temporary Enamel Demineralization Effects
Oxidative whitening treatments compromise enamel integrity through temporary demineralization processes that extend beyond optical modifications. You’ll experience surface roughness changes as carbamide peroxide alters hydroxyapatite’s molecular structure, shortening P-O bonds and displacing calcium and phosphate ions from crystalline matrices. The outermost 10 micrometers of enamel softens, mimicking early-stage decay patterns with reduced microhardness measurements.
Your enamel’s appearance transforms as mineral loss creates visible structural alterations—smooth surfaces become irregular, and tooth edges develop jagged characteristics. Accelerated lesion formation occurs after 72 hours of continuous exposure, with demineralization depth correlating directly to treatment duration. These chemical modifications extend progressively from surface layers into deeper enamel structures, creating temporary vulnerabilities to mechanical damage while oxidative agents remain active within the dental environment.
Understanding Sensitivity and Safety Considerations
While professional teeth whitening delivers dramatic aesthetic results, you’ll need to understand the sensitivity and safety considerations that accompany peroxide-based treatments. Transient enamel sensitivity occurs when hydrogen peroxide rapidly breaks down, releasing oxygen molecules that can temporarily affect pulp tissue.
You’ll minimize these effects through proper dosage concentration monitoring—professional treatments use 35-40% hydrogen peroxide in controlled 15-20 minute sessions, reducing pulp toxicity while maintaining ideal results. The American Dental Association Council on Scientific Affairs emphasizes that professional oversight guarantees safe outcomes.
At-home products contain lower concentrations, typically 10% carbamide peroxide (equivalent to 3.6% hydrogen peroxide), providing you greater control over application frequency. LED or laser activation accelerates peroxide breakdown, requiring careful time management to prevent excessive sensitivity while achieving your desired whitening goals.
Frequently Asked Questions
How Long Do Teeth Whitening Results Typically Last After Oxidative Treatment?
Professional oxidative whitening typically lasts 1-3 years, though you’ll experience whitening effectiveness variations based on your dietary habits and oral hygiene. Tooth sensitivity concerns shouldn’t deter maintenance treatments every 6-12 months to preserve your ideal results.
Can Oxidation Whitening Work on Dental Crowns, Veneers, or Fillings?
No, oxidation whitening won’t work on your dental restorations. Crowns, veneers, and fillings lack organic chromogens that peroxide targets. While oxidative whitening effectively lightens stained natural teeth, these inert materials resist oxidation, creating potential shade mismatches you’ll need to address.
Are There Foods or Drinks to Avoid During Whitening Treatment?
Verily, you’ll want to avoid tooth staining foods and limit beverage consumption during treatment. Coffee, red wine, dark sodas, berries, and acidic items compromise oxidation efficacy by depositing chromogens onto enamel, thereby reducing your whitening outcomes considerably.
How Soon Can I Repeat Whitening Treatments if Results Fade?
You can repeat professional whitening treatments every 6–12 months for maintaining whitening results. Timing of repeat treatments depends on your enamel health and lifestyle factors. Consult your dentist to establish a personalized schedule that preserves tooth integrity.
Does Oxidation Whitening Work Equally Well on All Natural Tooth Colors?
Yellow teeth lighten approximately 2-3 shades faster than gray tones during oxidation. Individual tooth response varies based on baseline chromogen density and tooth shade variations, meaning you’ll achieve more predictable results with yellowish discoloration than grayish hues.